Solid or acuqeus precipitate chemistry12/20/2023  
The formation of a solid precipitate is the driving force that makes the reaction proceed in the forward. A precipitation reaction is a chemical reaction which produces a precipitate when. We can use solubility rules to predict whether a precipitation reaction will take place. A precipitate is an insoluble 1 solid that forms when solutions are mixed. The solvent and soluble components of the reaction are called the supernatant or supernate. 
Thus precipitation reactions are a subclass of. The insoluble product compound is called the precipitate. A precipitation reaction is a reaction that yields an insoluble producta precipitatewhen two solutions are mixed. 3 A student tests two solids, solid C and solid D. Consider, for example, mixing aqueous solutions of the soluble compounds sodium carbonate and calcium nitrate. Modified by Joshua Halpern ( Howard University) 6.3: Precipitation Reactions is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. (h) Aqueous sodium hydroxide reacts with aqueous barium chloride to form a white precipitate of. 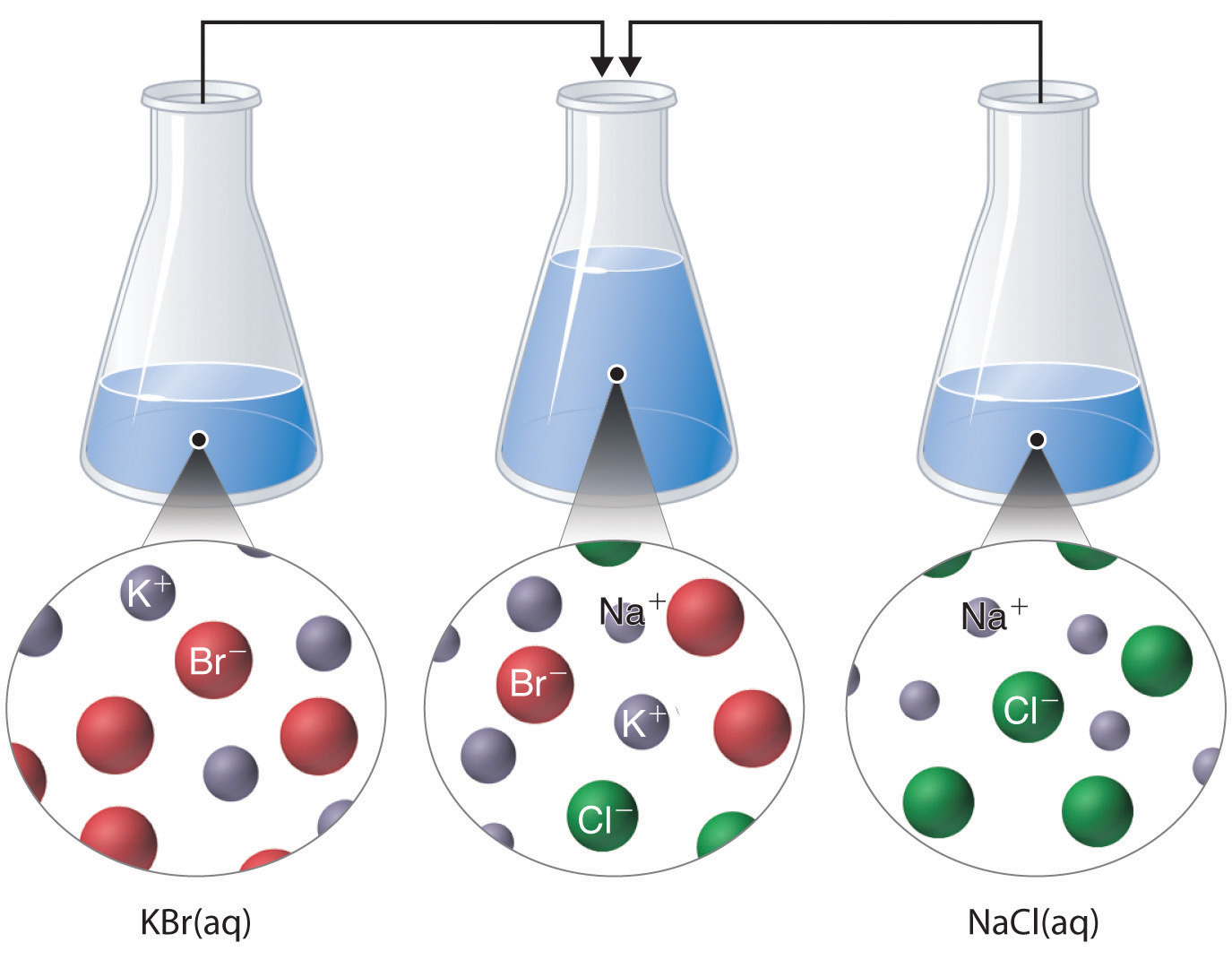
Characteristics: Manganese is a gray or reddish-white metal. Many reactions of this type involve the exchange of ions between ionic compounds in aqueous solution and are sometimes referred to as double displacement, double replacement, or metathesis reactions. It is important to realize that this equilibrium is established in any aqueous solution containing Ca 2 + and CO 3 2 – ions, not just in a solution formed by saturating water with calcium carbonate. Most common oxidation states: +2, +7 +3, +4, and +6 also exist. A precipitation reaction is one in which dissolved substances react to form one (or more) solid products. The insoluble solid, called the precipitate, initially forms a suspension, meaning that it is well dispersed in solution. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
